Description
Product Details
- EN 14683:2019 + AC:2019 TYPE IIR
- Surgical / Medical
- Our TYPE IIR masks are classified as surgical masks because of their great performance and they can be used surgical, medical areas or in public.
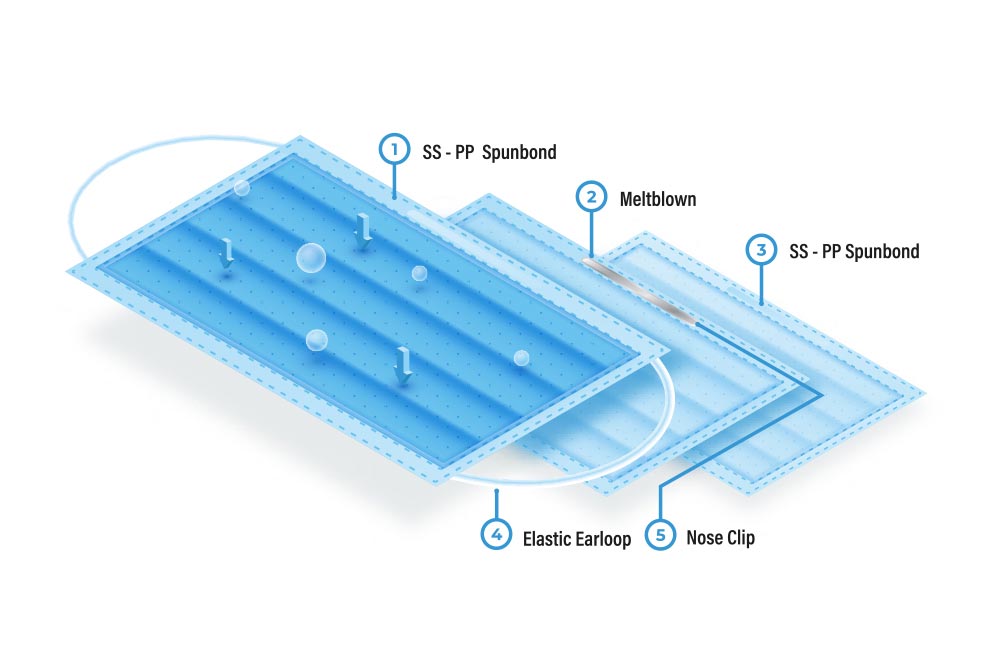
- 3 Ply / 3 Layers (SMS)
- Inner layer is made of soft spun-bond material for more comfort, soft absorbent, and hypoallergenic non-woven fabric.
- Middle layer is made of melt-blown filter non-woven fabric. Melt-blown material is the filter of your mask and gives you over 98% Biological filtration efficiency.
- Outer layer is made of spun-bond non-woven fabric and provides a barrier for splashes coming towards your face or going out from you.
- Disposable
- Our masks are for single use. Change it when you it gets damp or when you change environment.
- Splash Tested SRP > 20 kPa
- BFE > 98%
- Ultrasonic
- Materials stitched together by ultrasonic machine for better protection.
- Nose Wire
- Polypropylene coated soft and adjustable nose wire shapes around your nose makes your face covered better.
- Earloop
- Latex free comfortable round elastic, fits almost all adults
- Breathable
- Non-woven material lets your breathing easier.
- Non-Sterile
- Non-Woven
- Latex-Free
- Lint-Free
- Fog-Free
- When fit correctly on your face it doesn’t make your glasses foggy.
Material
- Pons Guard® Surgical / Medical Face Masks are made of 3 layers.
- Outer layer is made with Spun-Bond non-woven fabric.
- Middle layer is made with Melt-Blown polypropylene.
- Inner layer is made with Spun-Bond non-woven fabric.
- Our face masks have the following performances on tests made according to related European standards.
- Bacterial Filtration Efficiency (BFE) over 98%.
- Breathability Differential Pressure below 20Pa/cm2.
- Microbial Cleanliness below 20 cfu/g.
- Splash Resistance Pressure over 20 kPa .